Stent Application Micro-CT Study
Computed Tomography for Medical Devices
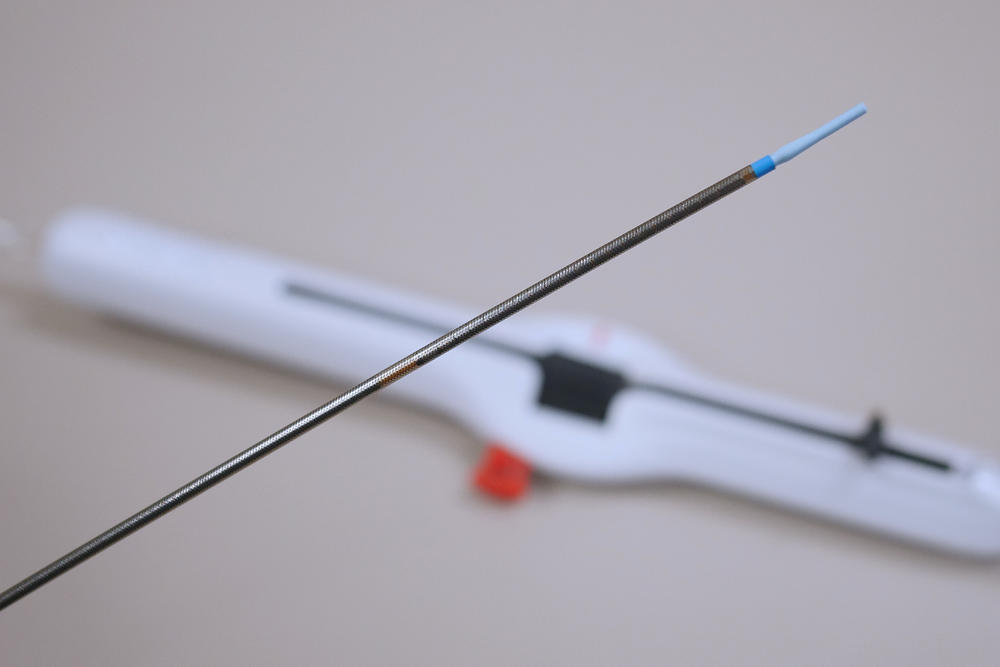
As a life saving device, stent engineering and development accuracy is crucial to the overall functionality of the device. Not only do medical device manufacturers need to verify the quality and function of the stent itself but they also need to consider the precision of the delivery device used to implant the stent into the artery.
Most stents are made up of a nitinol and platinum metal. This metal structure is squeezed down in a delivery device and placed into an artery. Once inside the artery, the stent expands out to open up the blockage and hold the structure together so the blood can flow freely and the body can heal.
When imaging a stent, most medical manufacturers are looking at the intersection points where the nitinol wire crosses the other to understand and characterize the wire wear that may occur over time. Manufacturers are also looking for any wire fracture or defects. Stents are manufactured using etching or laser cutting so it’s important to measure the accuracy of the cuts and identify any protruding burs or nicks that could cause harm to a patient or create stress risers that will fail in service.


Using X-ray to examine the delivery devices, a manufacturer can see the inner and outer details of the handle, cables, small laser welds, and plastic injected molded parts of the delivery handle. With 4D X-ray technology, the stent deployment action can also be seen as if it were being directly inserted into the artery.

Each stent does not have the same exact shape or flexibility. After CT scanning the stent, it is possible to perform a finite element analysis using the real world data set from the CT scan. This FEA software uses a simulated computer program to see if the stent twists or bends too much and if it will break or fail with its level of flexibility.
With the introduction of new stent materials in the marketplace, like absorbable stents, CT scanning proves to be an effective nondestructive testing technology when failure is not an option.
